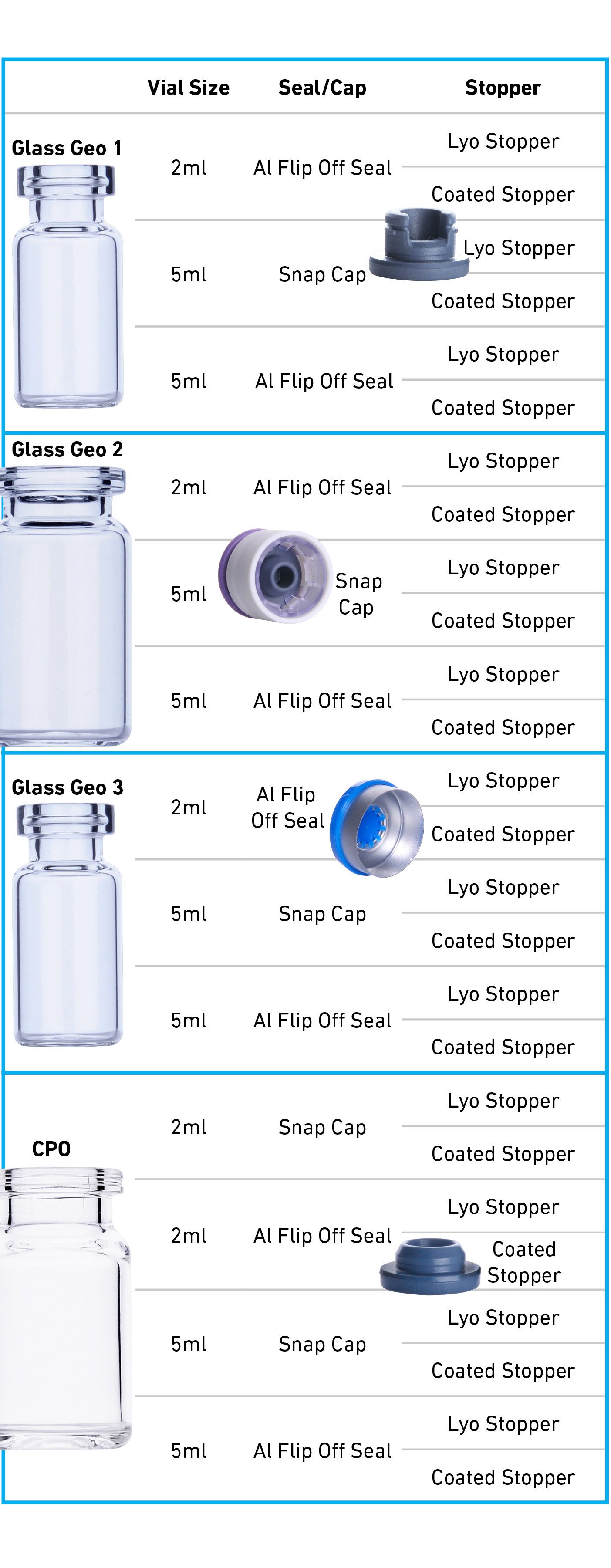
21 CFR Part 211 Subpart E - Control of Components, Drug Product Containers and Closures - LearnGxP: Accredited Online Life Science Training Courses

Visual Inspection of Parenteral Products Part 5: Container Closure Integrity - From Regulations to...
21 CFR Part 211 Subpart E - Control of Components, Drug Product Containers and Closures - LearnGxP: Accredited Online Life Science Training Courses