Foundation Medicine Acquires Lexent Bio, Inc., to Accelerate Liquid Biopsy Research and Development, and Advance Cancer Care

Foundation Medicine to launch liquid biopsy companion diagnostic following FDA approval - MedCity News

Foundation Medicine Eyes Liquid Biopsy Expansion, DNA Methylation Adoption, with Lexent Bio Acquisition | Inside Precision Medicine

Foundation Medicine on X: "Learn from a panel of our Foundation Medicine colleagues in part 2 of the “Liquid Biopsy in Clinical Practice” webinar on Tuesday, November 29th at 10:00 a.m. ET.
Roche France, Foundation Medicine and the Institute Gustave Roussy announce unique partnership to provide in-house liquid biopsy genomic testing to cancer patients in France | Gustave Roussy

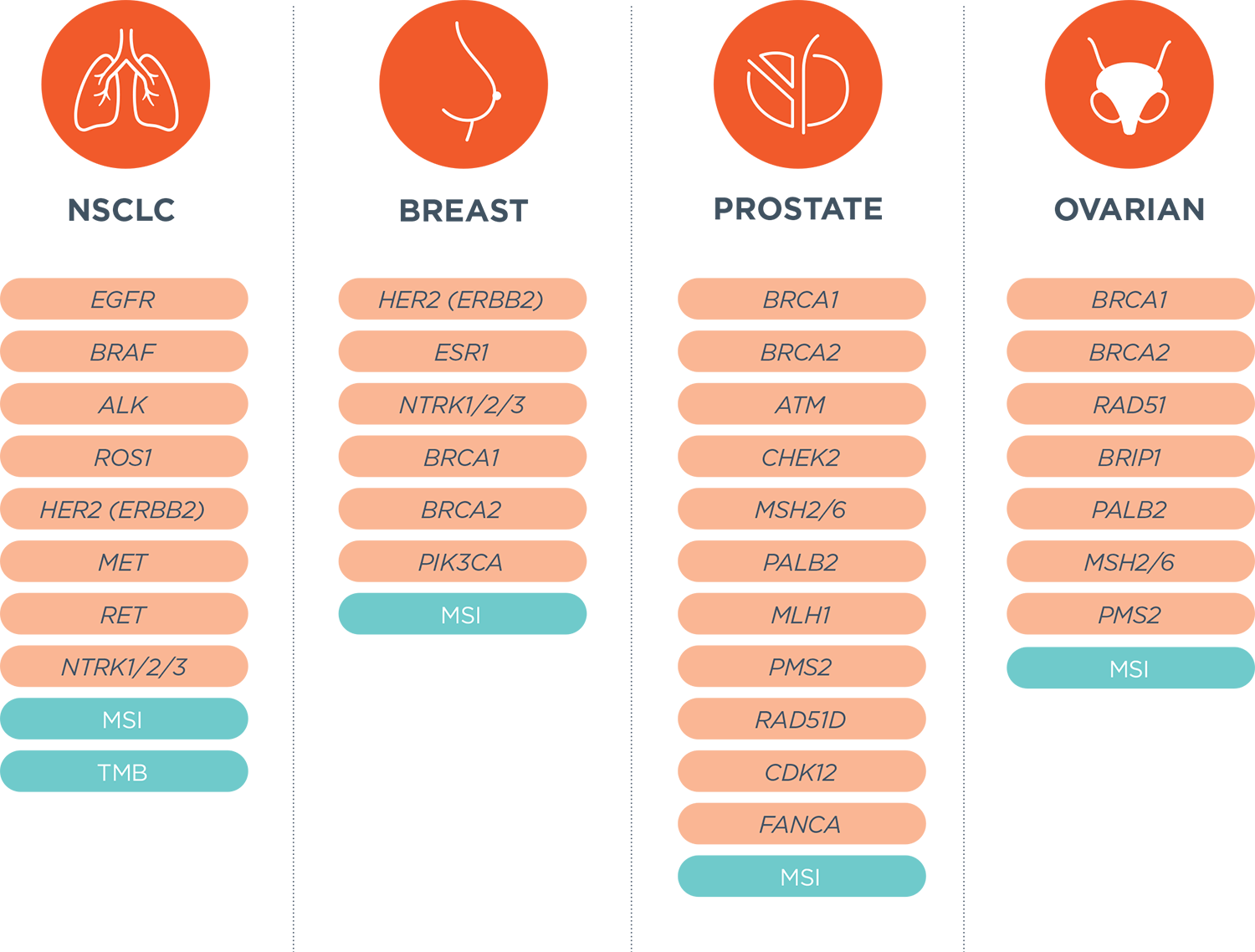
Liquid Biopsy to Identify Actionable Genomic Alterations | American Society of Clinical Oncology Educational Book

![Foundation Medicine review - 7 facts you should know [DEC 2021] Foundation Medicine review - 7 facts you should know [DEC 2021]](https://nebula.org/blog/wp-content/uploads/2021/12/FoundationOne-CDx-testing-kit.png)